The Plerixafor Injection Market is poised for steady growth at a CAGR of 7.2% over 2023-2031 to reach US$ 1,011.5 million by 2030. Plerixafor injections like Mozobil are used to mobilize hematopoietic stem cells in the blood for harvesting before autologous stem cell transplant procedures in blood cancer patients. Market expansion is driven by the rising prevalence of target conditions like non-Hodgkin's lymphoma and multiple myeloma globally. Over 334,000 people are estimated to be diagnosed with these hematologic malignancies worldwide annually.
North America will lead market growth through 2030, aided by supportive reimbursements for autologous HSCT protocols under private and public health plans in the US. Europe follows closely behind. Asia Pacific will display the fastest growth at a 9.8% CAGR between 2023-2030 owing to improving diagnosis rates and healthcare infrastructure. Key market players like Genzyme, Cadila Healthcare, Cipla, and Dr. Reddy’s Laboratories are launching branded and generic plerixafor injections across regions to expand affordability and access. Recent developments like Biocon’s biosimilar Myleukax launch in India, Genzyme’s regulatory approval of Mozobil in China, and Dr. Reddy’s USFDA authorized generic Mozobil launch signify rising industry focus on addressing demand across untapped markets.
Drivers:
- The rising target patient pool with annual incidence of non-Hodgkin’s lymphoma and multiple myeloma expected to increase by 12% and 18% respectively by 2030 will boost demand for plerixafor mobilization for HSCTs.
- Advancements and increased success rates of autologous HSCT procedures have led to greater physician preference for the same, driving plerixafor injection uptake. Over 85,000 autologous HSCTs were conducted globally in 2022.
Trends:
- Investments to expand plerixafor injection manufacturing capacities are rising significantly, with many generics players announcing facility upgrades recently to meet increasing industry demand.
- Strategic mergers and acquisitions between leading biopharma companies to enrich their oncology product portfolios and align with growing stem cell therapy needs also mark key trends impacting market outlook.
Opportunity:
The opportunity to tap into underpenetrated developing markets by improving awareness regarding role of plerixafor injections presents growth avenues worldwide.
- North America accounted for over 57% market share in 2023, aided by Genzyme, Mylan N.V., and Dr. Reddy’s strong regional presence coupled with increased product uptake following recent generic launches. Favorable reimbursements are boosting treatment rates.
- Europe held the second largest share of 30% in 2023, with prominent companies like Sanofi, Cadila Healthcare, and Sun Pharma recording rising sales across countries like Germany, France with growing demand for biosimilar plerixafor options opening avenues.
- Leading companies operating in the global plerixafor injection market are Genzyme Corporation, Sanofi S.A., Cadila Healthcare, Cipla Ltd., Dr. Reddy's Laboratories, and Biocon Ltd. among others. These players are focusing on new product launches, production capacity expansion, and strategic acquisitions to consolidate positions across key markets.
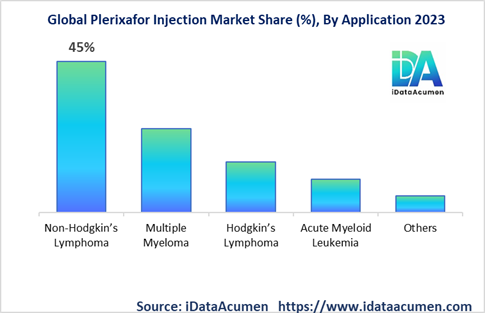
Market Segmentation:
- By Type
- Prefilled Syringe
- Injectable
- Lyophilized
- By Hematopoietic Stem Cell Transplantation Type
- Autologous
- Allogeneic
- By End-user
- Hospitals
- Specialty Clinics
- Stem Cell Banks
- Others
- By Combination Therapy
- Plerixafor + GCSF
- Plerixafor + Chemo + GCSF
- Others
- By Application
- Non-Hodgkin’s Lymphoma
- Multiple Myeloma
- Hodgkin’s Lymphoma
- Acute Myeloid Leukemia
- Others
- By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
Definition:
“Plerixafor injection is a hematopoietic stem cell mobilizer medication used to mobilize hematopoietic stem cells from the bone marrow to the peripheral blood for collection and subsequent autologous transplantation. It helps collect high numbers of cells efficiently in cancer patients eligible for transplantation procedures. The drug specifically blocks CXCR4 chemokine receptors on stem cells allowing them to move from bone marrow to blood circulation, from where they are eventually harvested. Plerixafor is majorly used in combination protocols with GCSF for satisfactory mobilization. Key target patient groups are non-Hodgkin’s lymphoma and multiple myeloma cases planned for autologous HSCT."